Current Research areas
Hard and soft tissue mechanical properties characterization
Bones are kind of rigid organs that constitute part of the endoskeleton of vertebrates. Primary function of the bone is to support and protect the various organs of the body, produce red and white blood cells and store minerals. Bone tissues are dense connective tissue. Bones have a variety of shapes and have a different internal and external structure, are lightweight but strong and hard, and they serve multiple functions.
The basic tissue of bone is osseous tissue, is hard and lightweight composite material. It is mostly made up of a composite material incorporating the mineral calcium phosphate in the form of calcium hydroxylapatite (it gives bones their rigidity) and collagen, an protein which improves fracture resistance (give elasticity).
We are trying to characterize both hard and soft tissue properties from macro to micro and nano scale.
Finite Element Analysis: A new clinical tool to assess boneQuality
We are working on to measure bone quality of osteoporosis patient. Osteoporosis diseases is now a days second most important health care problem, ranked by World Health Organization.
By using Finite Element Tool, we are measuring bone quality. In this study we are simulating the bone behaviour at different type of physiological loading condition. This study will be used to doctor for early prediction of osteoporosis induced fracture and effect of different medication on bone strength with time.
This research is conducted in collaboration with group and expert of Post Graduate Institute of Medical Education and Research Chandigarh.
To design of closed loop blood pressure control
Indian Institute Of Technology, Ropar has joined hands with PGIMER, Chandigarh to bring a new innovation to the healthcare facilities of India. We are currently working on designing a feedback controller that will automatically control the Mean Arterial Pressure of an anesthesized patient during a surgical operation by infusing the appropriate dosage of the corresponding drug. So far we have been successful in interfacing the syringe pumps (through which the drug is infused into the patient) with LabVIEW serially and control the action of the syringe pump. Now we are into the work of designing a fuzzy logic controller that would be able to regulate the mean arterial pressure by delivering proper dose of the drug through its control rule mechanism. Our earlier approaches were to design a state space controller for the same and validate its performance. Due to its few drawbacks we have shifted have research to fuzzy logic controllers.
Characterization of the mechanical properties of soft skin
The characterization of mechanical properties of skin is performed in various fields such as skin grafting, dermatology, impact biomechanics and forensic science. Currently we are performing two types of studies on the soft skin sample. First study involves the utilization of uniaxial & biaxial setup, which is integrated with digital image correlation (DIC) to obtain the material constants and stress-strain curves for skin. Second study includes the bulging of the skin to determine its mechanical properties, in this study we fix the skin sample on a control volume setup (developed in our lab) and skin bulging effect is produced by introducing the fluid inside the setup through syringe pumps.
Cell Mechanics and Mechanotransduction
The collective cell migration is responsible for tissue development, wound healing, cancer metastasis, homeostasis and many other biological processes. Understanding the dynamics of an epithelial cell monolayer will help to uncover the basic mechanism involved in the collective cell migration taking place in many biological processes.
We have well established cell culture room. We are currently culturing A549 & stably transfacted MDCK epithelial cell lines. An inverted fluoroscent microscope is employed to perform live cell microscopy to study the dynamics of epithelial cell monolayer.
In this current study we have assumed every point of epithelial tri-cellular junction (vertex) as a molecular particle & tracked (Fig. 1a) their location for the duration of 12 hours. Since the movements of two particles diffusing at molecular level are independent of each other, therefore vertices of monolayer which do not share common cells are considered for this study. The Mean square displacement (MSD) of the tri-cellular vertices plotted against time (Fig. 1b) give the probability of finding that tricellular vertices at that particular instant of time.
Biological material characterization
Biomechanical characterization is our one of the active research area where we are extracting the biomechanical properties of biological materials in-vitro. To perform a smooth biological function for a healthy human body the intrinsic biomechanical properties play an important role. We have facility to extract the mechanical properties of biological sample from small scale to bulk. The knowledge of mechanical properties of hard and soft biological material such as bone, cartilage, tendon, artery, skin also keratinized epithelial tissues such as nail, hair are useful to predict the effect and progression of disease and also the modeling and designing of the replacement material which are having whose market all over the world.
The mechanical properties we are studying such as L-D curve, hardness, modulus, tensile and compressive properties, bending, dynamic mechanical properties and damping.
Nano-mechanical characterization of De-mineralized Bovine Bone
Bone derives its mechanical properties from a complex arrangement of collagen type I fibrils reinforced with nano-crystalline apatite mineral in extra-and intra-fibrillar compartments. De-mineralized bone represents organic matrix containing individual collagen fibrils which has been investigated for mechanical properties in the present study. Elemental composition analysis through Energy Dispersive X-Ray Spectroscopy (EDX) has been used to assure complete removal of hydroxy-apatite from bone during de-mineralization for probed depth ≈ 2 μm. The effect of load magnitude variation on the measured mechanical properties has been examined through nano-indentation for loads ranging up to 1000 μN. At low loads, the indentation modulus of the organic matrix was ≈ 20% greater than at higher loads. As the ratio of indentation depth to surface roughness is increased, the variability in material properties decreased substantially. Modulus magnitudes obtained from dynamic indentations (constant frequency of 30 Hz) have been used to incorporate influence of strain rate on material properties and to make its comparison with quasi-static indentations. Modulus magnitudes have been calculated as 4.5 GPa and 2.5 GPa respectively for dynamic and quasi-static indentation. These findings are relevant for understanding the bio-mechanics and design principles of collagen matrices in calcified tissues.
|
|
|
|
|
|
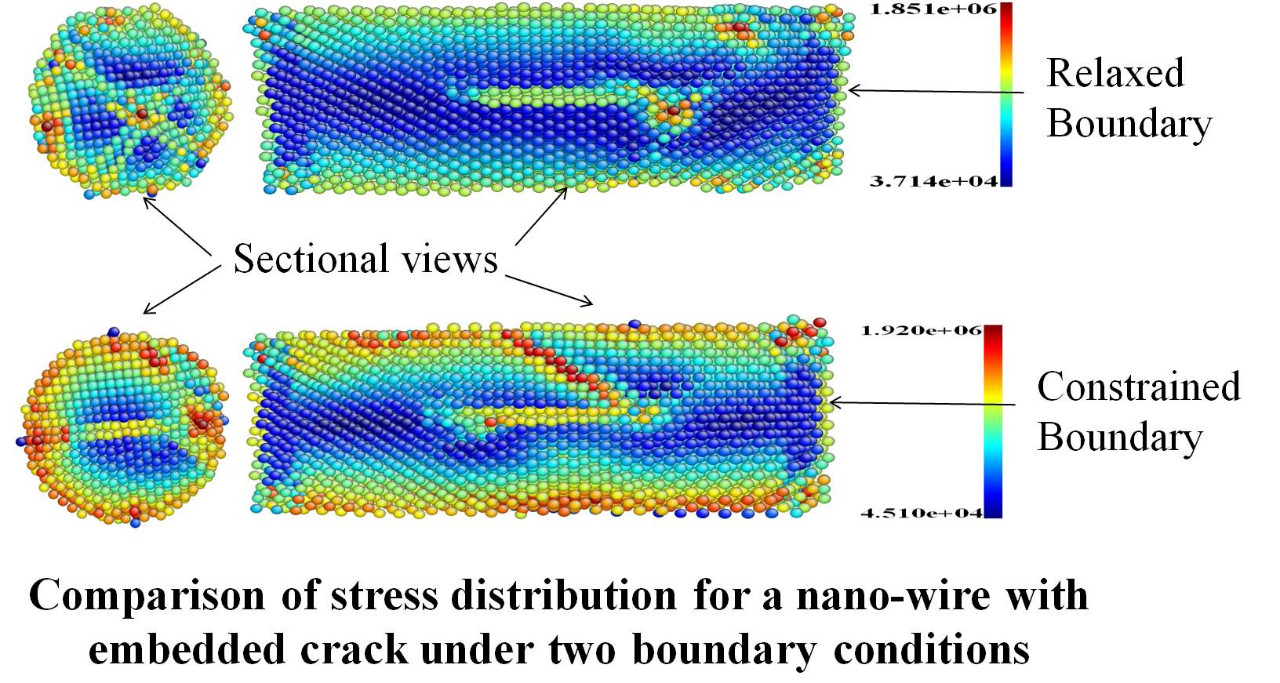
Characterization and Modeling of Nanowires and Nano Composite
Nano-wires have a wide range of potential applications, such as in nano- electromechanical systems (NEMS), as a active components of circuits, sensors and actuators in NEMS, optical sensors, resonators and force sensors, high-resolution scanning probe microscope (SPM) tips, and reinforcements for nanocomposites.So detailed studies regarding the properties of materials are required at scaled down dimensions before going for manufacturing systems using these nano-wires. However, when the metallic components are scaled down to such small sizes, it is not easy to control the process of manufacturing the nano-wire. There is a great proba- bility of getting defects in the nano-wires. So one cannot ignore the presence of defects. When materials are bent or twisted, they experience strain gradients which are known to alter the plastic response of the materials. We are also working on providing techniques to accurately and quickly characterize nanomaterials, so that we are able to evaluate different nanomaterials for designing high performance nano-systems.
Nano mechanical Characterization and Modeling of Animal Bones
To investigate the mechanical behavior of bone, dentin using nano- and micro-indentation techniques. The development of numerical and semi-analytical models (at multiple scales) for estimation of mechanical properties, and for correlation with experimental data, is also considered a key element of this research.
In fact, nanoindentation is one of very few techniques which can be used to characterize biomaterials in their native environments, e.g. being immersed in liquid. Moreover, the fully understanding of the properties of bone at the nanoscales will provide important new knowledge for the development of target-specific therapeutic treatments on altering the bone microstructure to prevent bone fracture, which would have great impact on improving human health.
Nanomechanical Characterization and Modeling of Bio-systems
Mechanical properties of the bio materials changes with biological processes involve. To quantify cellular mechanical behavior, we need to measure the displacement response of bio-systems to an applied force. The characterization of compliant cellular structures requires nano-Newton and nanometer resolutions. AFM and Nanoindenter are suitable to image and characterize the behavior of intrinsic/natural biological processes without much influence on the processes due to the delicate testing forces. The two techniques can be also applied to measure mechanical behavior deep into the cell membrane.
Nano Indentation Images:

Bone indent 3d
Indent image of bone
Indent On Alumina witm 500µn force
Research Area:
Research work includes determination of mechanical characteristics of metals (defected gold nano-wires), biological materials (proteins) and nano-composites (polymer-CNT) using atomistic modeling and experimental techniques. Atomistic Modeling is a very good tool to predict the mechanical behavior of materials at very small length and time scales. It uses the interactions among the basic constituents (atoms, molecules etc.) of the materials by defining suitable potential function. Potential function defines energy landscape as function of separation between atoms. Potential function varies from material to material. This potential function can be used to find the force acting between atoms of a particular material under specific deformation and hence material properties can be defined. He is also studying nano-indentation (in particular) to determine material properties and its application to metals, biological materials and nano-composites.
Applications:
Atomistic modeling technique is very useful in determining the properties of materials at very small length and times scales which are very hard or sometimes, nearly impossible to determine using experimental techniques. So characterization of metal nano-wires can be very useful in fabricating materials for NEMS (Nano-Electro-Mechanical Systems) etc.. Understanding the biological materials behavior can provide great insight to design materials for their replacements in implants and for producing medicines for pharmaceutical industry. Characterizing nano-composites can very useful in designing high strength materials for Defense, Aircraft, Automotive applications etc.
Bone is a complex tissue that not only provides structural support and movement to our body, but also has important functions in blood production and in regulation of various minerals and growth factors. Bone’s structure and function are primarily maintained by three cell phenotypes - Osteoblasts, Osteocytes and Osteoclasts. Osteoblasts and Osteocytes are responsible for bone formation and mechanosensation, respectively, while Osteoclasts absorbs bone matrix to facilitate bone repair and remodeling. The network of Osteocytes within bone is believed to sense mechanical stimuli that are further communicated to Osteoblasts and Osteoclasts leading to bone adaptation to the mechanical environment, the process known as Mechanotransduction. In vivo studies have shown that bone is responsive to mechanical parameters such as strain magnitude, strain rate and strain gradients. These mechanical stimulations typically lead to new bone formation at some specific skeletal sites; however the quantitative dependence of new bone formation on the mechanical environment is not well-understood. Our approach is basically to study the site specific differences in long bone cross-sectional adaptation. We are trying to develop the algorithm by which final geometry at any cross-section of long bone can be predicted after a certain mechanical stimuli protocol